Raising the standard in chemical burn first aid
WITHIN TEN seconds, chemicals can begin penetrating tissue. The next 50 seconds may determine whether the injury remains reversible. Beyond that, the risk of deeper tissue damage, extended recovery and hospital admission increases significantly.
The difference between a minor incident and a life-changing injury often comes down to what happens in that first minute.
Across the UK, most facilities rely on emergency washing equipment installed in accordance with EN15154. The Diphoterine® systems conform with EN15154 Parts 3 and 4.
But compliance is the baseline.
The real question is: does your response strategy control the outcome?
Water: Conventional, but it has limitations
Water has long been the default response to chemical incidents. High-flow emergency washing equipment rinse and dilute chemicals from the surface of the tissue. When accessed quickly, this can reduce surface concentration.
However, water is passive. It dilutes externally present chemicals but does not actively address substances that have already begun penetrating tissue, a process that typically starts within 10 seconds.
Chemical injuries progress through stages: Contact, Diffusion and Chemical Reaction. Once diffusion begins, the aggressor can continue moving into deeper layers, and water cannot reverse this process.
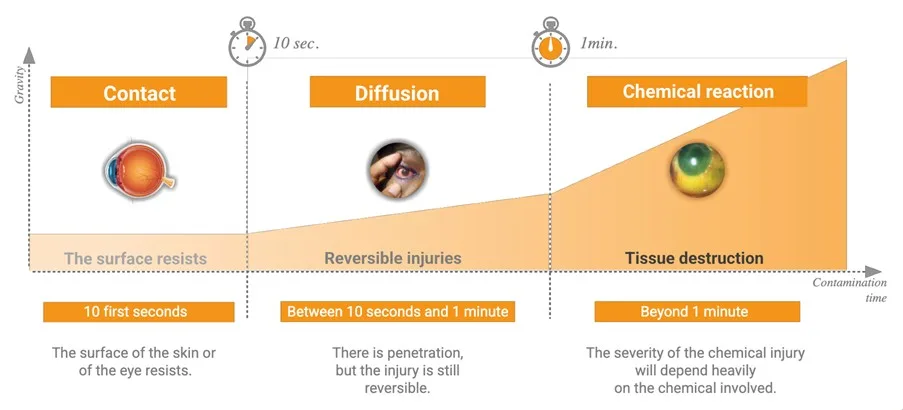
Water is hypotonic, meaning it has a lower solute concentration than tissue. This can allow fluid movement into tissue, potentially assisting contaminant spread.
There are also practical considerations. Emergency washing equipment is fixed in place, may not be reachable within 10 seconds, require maintenance and legionella management, and use approximately 900 litres for full-body decontamination.
Water meets the standard, but it has limitations.
Saline: Gentler, same principle
Saline is commonly used for eye irrigation because it is isotonic (having a similar salt concentration to the body) and therefore unlikely to push chemicals further into tissue. It was primarily designed to relieve minor irritation and flush away small foreign particles or contaminants. Its mechanism, like water, remains rinsing and dilution.
Saline does not bind chemical aggressors or limit ongoing diffusion.
Buffer Solutions: Focused on pH Control
Buffer solutions are formulated to stabilise pH of the tissue within a defined range. They are typically composed of weak acid–base pairs and are widely used in laboratory and pharmaceutical applications to stabilise pH.
In the context of chemical incidents, buffers act by moderating acidity or alkalinity at the surface of the tissue.
Although buffers are intended to stabilise pH, they are not engineered to remove chemicals that have already diffused into tissue or to bind across the 7 major classes of chemical aggressors.
Diphoterine®: Active Chemical Decontamination
Diphoterine® was developed specifically for chemical splashes and represents a fundamentally different approach. As a Class IIa Medical Device, it is formally licensed and proven to perform as intended.
It is hypertonic and amphoteric. Its hypertonic nature creates an osmotic gradient that helps draw chemicals out of tissue, while its amphoteric properties enable binding of all irritant and corrosive substances.
Rather than simply rinsing the surface, Diphoterine® actively captures the aggressor, limiting further tissue interaction and helping return the affected area towards a safe physiological pH. By acting on both the chemical and the diffusion process, Diphoterine® reduces the likelihood of deeper tissue destruction, extended recovery time and hospital escalation.

Diphoterine’s® optimal response window is 60 seconds and it has also been shown to remain effective when applied up to 24 hours post-incident. Its portability allows it to be taken directly to the casualty rather than moving them to fixed emergency washing equipment. Decontamination can be completed within 6 minutes using approximately 5 litres for full-body exposure, reducing hypothermia risk compared with non-heated emergency washing equipment.
Users frequently report immediate pain reduction, supporting calmer casualty management and clearer clinical assessment.
Moving beyond minimum compliance
EN15154 establishes essential requirements for emergency washing equipment, but standards define the minimum, not the optimum.
Chemical burns account for a disproportionate share of severe and fatal injuries, and modern industrial chemicals are increasingly complex.
Water and saline rinse and dilute. Diphoterine® controls outcomes.
For organisations serious about protecting people and managing risk, downtime and liability, the first minute matters.
The question is no longer whether you are compliant. It is whether your response strategy controls the outcome.
Chemical first aid is evolving. Is your site keeping pace?
For more information, visit www.diphex.com
HSM publishes a weekly eNewsletter, delivering a carefully chosen selection of the latest stories straight to your inbox.
Subscribe here


